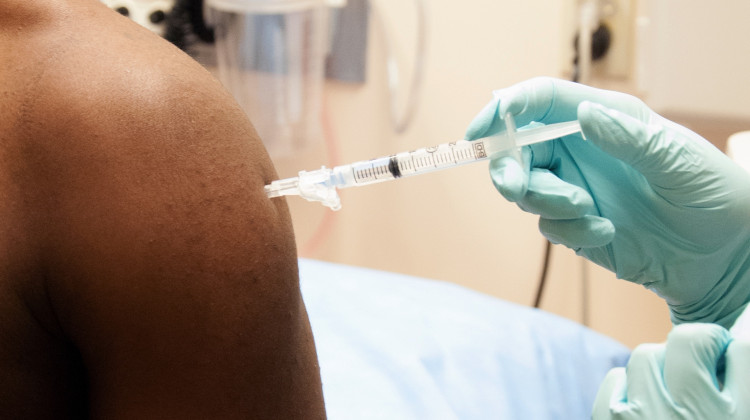
A man enrolled in a clinical trial receives a dose of the investigational NIAID/GlaxoSmithKline Ebola vaccine in 2014.
Photo courtesy of the National Institute of Allergy and Infectious DiseasesIn the U.S., Black people are twice as likely as White people to get Alzheimer’s disease, according to data from the National Institute on Aging. Yet Black patients are severely underrepresented in clinical trials for Alzheimer’s treatments. One study that began in 2016 to develop a drug to treat Alzheimer’s had only eight Black participants out of 813.
This is not unique to Alzheimer’s research. While 30 percent of all U.S. prostate cancer deaths are among Black men, they make up only 3 percent of participants in cancer clinical trials.
Health experts say this lack of representation leads to widening health disparities and ballooning health care costs.
An upcoming event in Indianapolis aims to increase awareness about the importance of diversity in clinical research. The “Better Journey to Health” event will take place from 5-8 p.m. on Sept. 14 at the Ivy Tech Community College Culinary and Conference Center.
The event is free and organized by the nonprofit Center for Information and Study on Clinical Research Participation. Panelists will answer questions like what to expect as a clinical trial participant and the benefits and risks of participation.
One of the panelists will be Lena Harvey, a 37-year-old Indianapolis woman who lives with sickle cell disease. Harvey will discuss her experience as a participant in clinical trials.
The lack of diversity is due in part to mistrust
While there are many reasons for lack of representation in clinical trials, Harvey said trust in the medical community is a big one.
Many of her older family members and friends do not interact with the health care system outside of emergencies or urgent problems because of the history of medical abuse. But she thinks that’s changing, as Black community members, especially young people like her, have access to more information than ever before.
“We are more connected than ever,” she said. “The internet absolutely changed everything. It offered us a more level playing field in terms of access to information.”
This includes access to support groups, patient advocates as well as information about clinical trials and specific doctors.
Harvey said she has taken a proactive approach to her health, empowering herself with information to make sure she got the care she needed. She participated in two clinical trials on drugs and procedures for sickle cell disease. Plenty of internet research and conversations with her husband and trusted sickle cell physicians helped her come to the decision to enroll. She understood that even if the trials did not help her personally, the results of the studies could help others down the road.
“Because sometimes it takes several years, decades for people working on something to get it to where it really needs to be. None of that work is in vain,” she said.
Why representation matters
Clinical trials are typically initiated by a research question from community organizations or scientists at academic centers or industry labs. Then, funding flows in from federal agencies, the private sector or charitable foundations. The clinical trial team then recruits participants through social networks or primary care settings. This is where more than half of the trials fail.
According to a report from the National Academies of Sciences, Engineering and Medicine, 55 percent of clinical trials stop because they were not able to recruit the participants needed to complete the study. This happens even after money and resources have been flowing into the trial for months or even years.
The financial fallout from lack of representation in clinical trials extends to individuals and their communities. Disparities for some health and quality-of-life outcomes, including life expectancy and years in the labor force due to physical ability, could be mitigated if clinical trials were more inclusive.
“Of course, better representation in clinical research will not completely alleviate these disparities — after all, they have many interconnected and interdependent causes. However, to the extent that representation in clinical research may improve generalizability of scientific findings across a range of clinical studies for these important health states … representative clinical research may play a role in alleviating these inequities,” according to the NASEM report.
If better representation in clinical trials would alleviate just 1 percent of health disparities, it would result in economic gains of as much as $40 billion for diabetes and $60 billion for heart disease alone, the report explains.
Treatment and prevention both depend on representative studies
Experts refer to a drug called warfarin as a cautionary tale of how lack of representation can be detrimental to patients’ outcomes.
Warfarin is used to prevent clots in blood vessels and is crucial to prevent deadly conditions like strokes and pulmonary embolisms. It is also a leading cause of adverse drug events in the U.S. because the wrong dose of warfarin can result in complications like excessive bleeding and even death.
Patients require different doses of warfarin, and that’s largely because of genetic factors. For example, patients of African ancestry require a higher average daily dose of the drug than patients of Asian ancestry. But most of the trials studying warfarin involved White people of European ancestry. Warfarin was approved for human use in 1951, but it took 62 years for scientists to learn that genotype-guided dosing is important for patient outcomes.
A lack of diversity in clinical trials can also negatively impact people of color when it comes to health screenings and other preventive measures.
Black adults in the U.S. have the highest rates being diagnosed with and dying from colorectal cancer.
Yet the U.S. Preventive Services Task Force, which makes recommendations for screenings that are oftentimes used by insurers to determine when screenings will be covered, said it was not able to make a specific, separate recommendation on colorectal cancer screening in Black adults due to a lack of representative studies, according to a report from the National Academies of Science, Engineering and Medicine.
“Unfortunately, we just don’t have the studies in these populations that allow us to say with certainty whether or how to adapt our prevention guidelines,” wrote Dr. Kirsten Bibbins-Domingo, a UCSF epidemiologist and biostatistician and one of the contributing authors on the report.
This story comes from a reporting collaboration that includes the Indianapolis Recorder and Side Effects Public Media, a public health news initiative based at WFYI. Contact Farah at fyousry@wfyi.org. Follow on Twitter: @Farah_Yousrym.
 DONATE
DONATE





 View More Articles
View More Articles




 Support WFYI. We can't do it without you.
Support WFYI. We can't do it without you.